Why Is There a Green Ring Around Your Hard-Boiled Egg Yolk? (And How to Fix It)
You are preparing a beautiful fresh salad, and you slice into your perfectly peeled hard-boiled egg—only to find an unappetizing, greenish-gray ring surrounding the yolk. It looks strange, it often has a slightly chalky texture, and it carries a faint sulfur smell.
Many people throw these eggs away, assuming they have gone bad. But the truth is entirely rooted in kitchen chemistry, not food safety. Here is the exact science behind the dreaded “green ring” and how to boil your eggs perfectly every single time.

What Causes the Green Ring? The Science of Sulfur and Iron
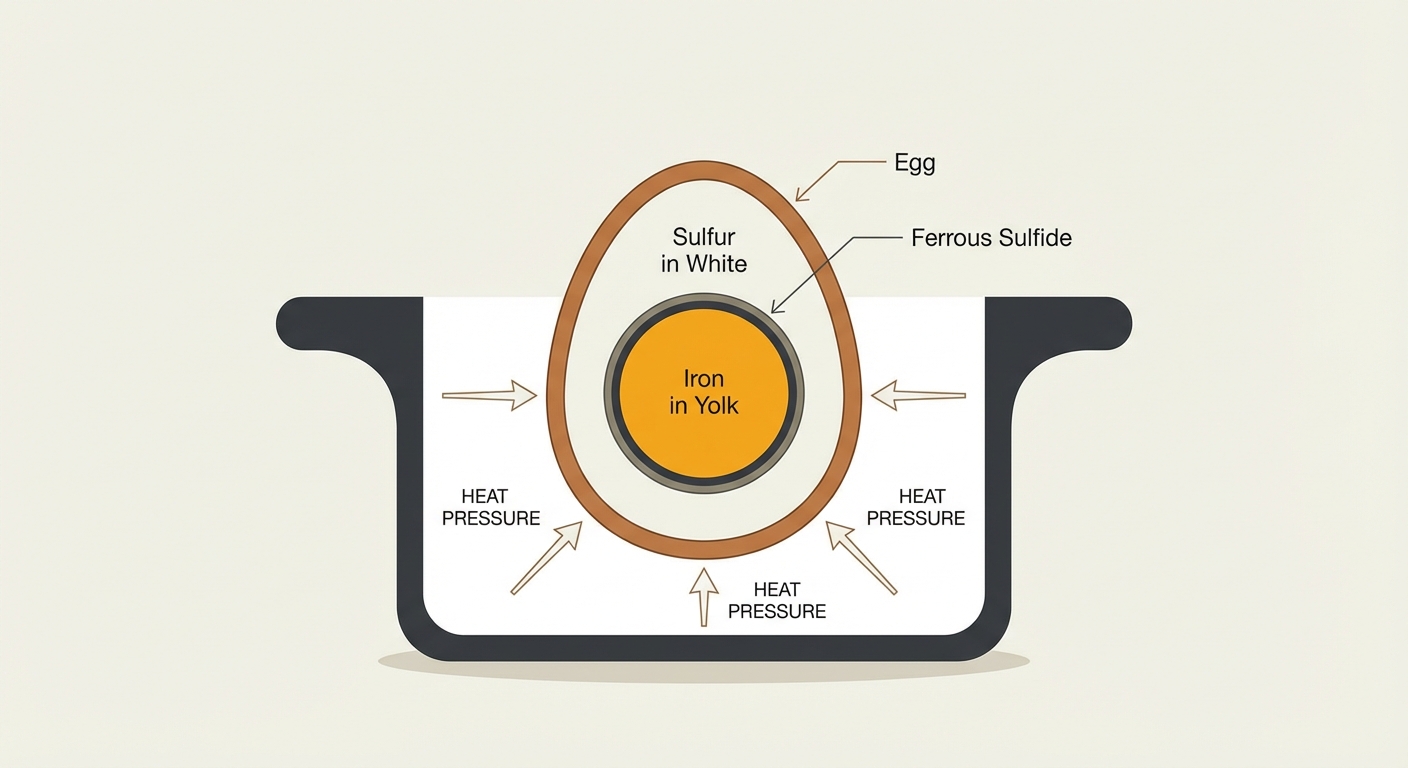
The green ring is the result of a harmless chemical reaction that occurs when an egg is exposed to too much heat for too long. Here is what happens inside the shell when you overcook an egg:
The Sulfur in the White
Egg whites naturally contain amino acids that are rich in sulfur. As the egg gets hot, these proteins break down and release hydrogen sulfide gas.
The Iron in the Yolk
The egg yolk is naturally packed with iron (a vital nutrient for your blood and brain).
The Chemical Collision
Heat creates pressure inside the egg, driving the hydrogen sulfide gas inward toward the cooler center (the yolk). When the sulfur gas hits the iron in the yolk, they react to form a new compound called ferrous sulfide. Ferrous sulfide is dark in color. When it sits on top of the bright yellow yolk, our eyes perceive it as that distinct greenish-gray ring.
Is It Safe to Eat?
Yes, absolutely. Ferrous sulfide is 100% harmless to ingest. The only downside to the green ring is that the egg is overcooked, meaning the yolk will likely be dry, chalky, and slightly rubbery.